Submitted by Administrator on Wed, 29/03/2017 - 11:46
Affiliate PI Peter Rugg-Gunn’s group at the Babraham Institute has mapped the changes in the three-dimensional shape of DNA that occur as human stem cells specialise towards becoming developing neural cells, the cells that make up the nervous system. These changes in DNA shape rewire the circuitry that connects genes with their control switches, which instruct them to turn on or off during neural development. These findings open up new ways to improve the efficiency of driving stem cells to become different types of cell, which is a prerequisite for many stem cell-based therapies. The research also provides new insights into which genes are inappropriately turned on or off in developmental disorders due to mutations in their control switches.
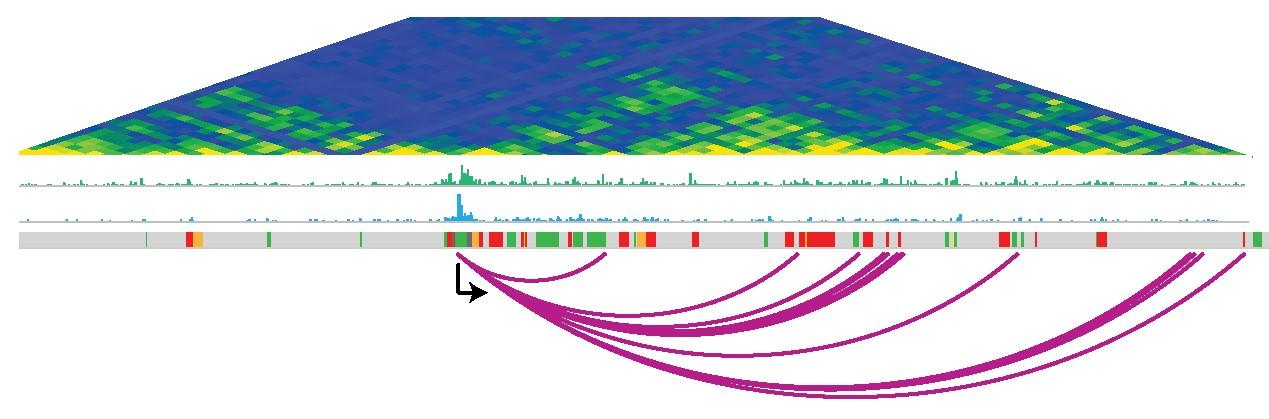
In the current research published in the journal eLife, the scientists set out to reveal how loops between genes and their regulatory elements change as human stem cells develop into neural cells. They mapped the loops between genes and their regulatory elements in unspecialised human embryonic stem cells and in stem cells in the process of developing into neural cells. The scientists uncovered widespread remodelling with the DNA loops being extensively rewired after the stem cells specialised into neural cells (i.e. loops were formed and lost). Also the regulatory elements were frequently toggled between “active” and “inactive”.
Read more: Babraham Institute
Publication details:
Freire-Pritchett P, Schoenfelder S, Várnai C, Wingett SW, Cairns J, Collier AJ, García-Vílchez R, Furlan-Magaril M, Osborne CS, Fraser PJ, Rugg-Gunn PJ, Spivakov M. Global reorganisation of cis-regulatory units upon lineage commitment of human embryonic stem cells. eLife 2017;10.7554/eLife.21926