Scientists at the Cambridge Stem Cell Institute have revealed new insights into cellular signaling processes that could improve chemotherapy treatments.
In research published in the journal Nature Chemical Biology, the study found that small signaling packages (known as extracellular vesicles) have the specific enzyme activity required to kill some cancer cells, without depleting essential nutrients required for healthy cell function.
The findings significantly extend our understanding of cell signaling biology whilst also having important clinical applications for cancer treatments.
The research team, led by Dr Stefano Pluchino from the Wellcome Trust – MRC Cambridge Stem Cell Institute, and Dr Christian Frezza from the MRC Cancer Unit, used stem cells to study the extracellular vesicles that are released to send signals into the surrounding environment.
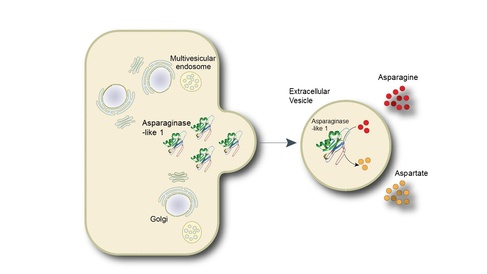
The scientists isolated these vesicles and measured their enzyme activity. The most active enzyme was found to be Asparaginase-like 1, a variant of the enzyme used in the treatment of acute lymphoblastic leukaemia, a blood cancer often diagnosed in younger people.
In the treatment of cancers, the chemotherapy enzyme works by depleting asparagine, a naturally occurring chemical in the body that cancer cells need to survive. However, the treatment also depletes a chemical called glutamine, which the healthy cells in the body need in order to function normally.
The scientists found that the activity of Asparaginase-like 1 enzyme in the isolated vesicles acted specifically on breaking down asparagine, and importantly, did not affect the levels of glutamine.
“Current drug treatments of acute lymphoblastic leukaemia presents a difficult balancing act: removing enough asparagine so that cancer cells cannot survive, but leaving enough glutamine to ensure normal cells in the body can thrive”, explain Dr Pluchino. “The discovery that the Asparaginase-like 1 in vesicles depletes asparagine but does not affect the glutamine could provide an alternative anti-cancer therapy that could limit side effects such as liver toxicity that can occur when glutamine is depleted”.
The discovery of this ‘clean-acting’ Asparaginase-like 1 enzyme and the development of technologies around this research forms the basis of a patent owned by Cambridge Innovation Technologies Consulting Ltd, of which Dr Pluchino is a Co-founder and Director. This research demonstrated the potential for the understanding of fundamental cell biology to be scaled up to improve human health.
Dr Pluchino and his team are based at the Wellcome Trust – MRC Cambridge Stem Cell Institute, a world-leading centre for stem cell research with a mission to transform human health through a deep understanding of stem cell biology.
The support of the European Research Council, Medical Research Council, the Italian Multiple Sclerosis Association, the UK Regenerative Medicine Platform Hub “Acellular Approaches to Therapeutic Delivery”, the Evelyn Trust and the Bascule Charitable Trust is gratefully acknowledged by Stefano and the team.
Publication details:
Extracellular vesicles are independent metabolic units with asparaginase activity. Nunzio Iraci, Edoardo Gaude, Tommaso Leonardi, Ana S. H. Costa, Chiara Cossetti, Luca Peruzzotti-Jametti, Joshua D. Bernstock, Harpreet K. Saini, Maurizio Gelati, Angelo Luigi Vescovi, Carlos Bastos, Nuno Faria, Luigi G. Occhipinti, Anton J. Enright, Christian Frezza, and Stefano Pluchino is published in Nature Chemical Biology. http://dx.doi.org/10.1038/nchembio.2422
