Dr Irving Aye
Placental metabolism and development
Departmental affiliation: Department of Obstetrics & Gynaecology
Biography
Irving completed his undergraduate and master’s degrees in Pharmacology at the University of Auckland NZ, followed by a PhD in Obstetrics at the University of Western Australia. He then completed post-doctoral fellowships in the USA (San Antonio and Denver) before joining the Dept of Obstetrics & Gynaecology, at the University of Cambridge as a Research Associate. In 2018, he was awarded the Next Generation Fellowship from the Centre for Trophoblast Research to study the role of fetal sex differences in placental function. In 2022, he was awarded the MRC career development award to start his laboratory investigating the role of metabolism during placental development.
Research Summary
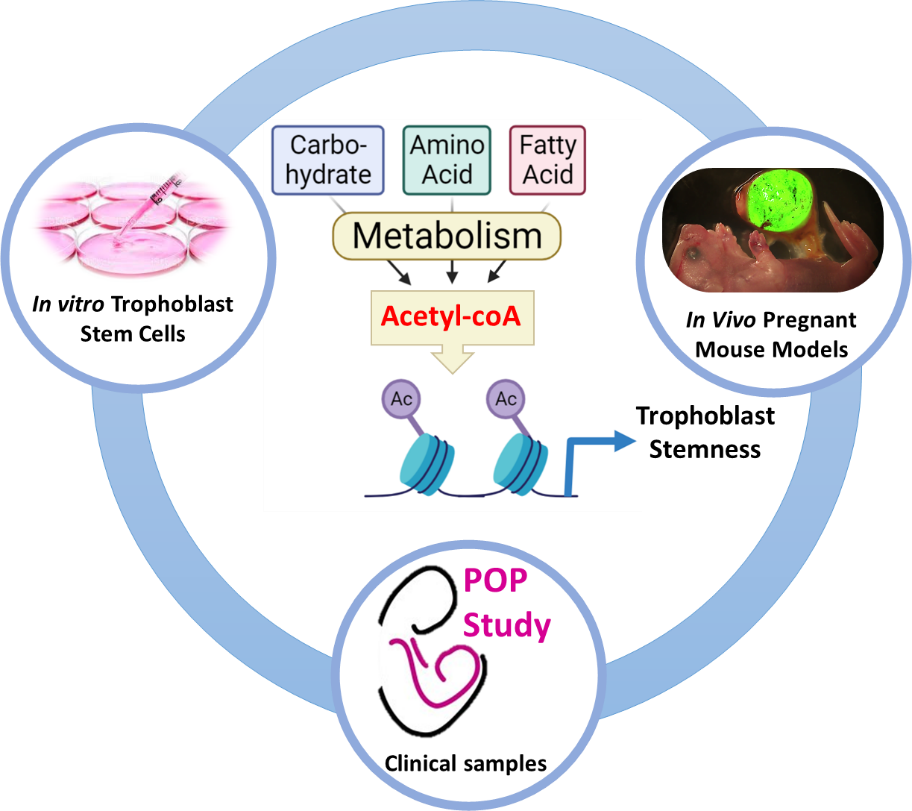
Many of the major pregnancy disorders originate from poor placental development in early pregnancy (i.e. the first trimester). However, there is little mechanistic understanding of how this occurs. The placenta's functional capacity is dependent on coordinating the self-renewal and differentiation of placental stem cells known as trophoblasts. Our long-term goal is to understand the interplay between metabolism and epigenetics during early placental development. We are currently examining the link between acetyl-coA availability and histone acetylation during trophoblast differentiation. Acetyl-coA lies at the intersection of carbohydrate, amino acid and fatty acid metabolism, and it is also the acetyl-donor for histone acetylation. Moreover, histone acetylation is highly sensitive to physiological changes in cellular acetyl-coA concentrations. We aim to define the mechanistic links between acetyl-coA metabolism and histone acetylation in regulating trophoblast fate and placental development.
A key feature of our research is the use of a variety of experimental models; including human trophoblast stem cells to investigate the molecular mechanisms, and pregnant mice to establish the physiological significance. Furthermore, analyses will be carried out using placental samples from a prospective cohort (Pregnancy Outcome Prediction Study) of normal and complicated pregnancies to determine the clinical relevance of our findings.