Research published this week in Science Advances by Dr Shota Nakanoh in a collaboration between Dr Teresa Rayon’s lab at the Institute and Professor Ludovic Vallier’s lab at the Wellcome-MRC Cambridge Stem Cell Institute has determined that the degree of cell crowding in the early human embryo influences whether cells develop as extra-embryonic cells or become a part of the embryo and eventually give rise to skin, hair and nails.
Combining the new culture method with three-dimensional culture and single cell RNA sequencing analysis, Dr Nakanoh, the lead researcher on the project, used human pluripotent stem cells (hPSC) to learn more about how amniotic and surface ectoderms arise in human embryos at around gastrulation (around two weeks after fertilisation).
The mystery of similar but different tissues – amniotic and surface ectoderms
Amniotic ectoderm is a single cell layer that forms the membrane surrounding the developing embryo. It also provides essential signals for human embryo development, however how the amniotic membrane comes about is still not fully known in humans.
Surface ectoderm is a dense sheet of epidermal progenitors which gives rise to the surface covering of the body, such as skin, and the associated features of teeth, hair and fingernails. Its artificial production is of medical interest if technologies to generate it could be improved.
Amniotic and surface ectoderm cells are specialised at an early stage of development and largely share biological features, such as gene expression patterns and signal requirements. It has been a mystery why these distinct tissues are so similar and how they differentiate during development.
Cell crowding affects cell identity
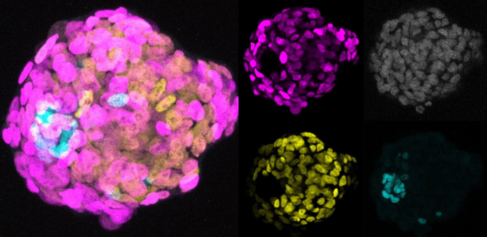
Dr Nakanoh customised culture conditions for hPSCs and found that the supplements Activin A, BMP4, a GSK3-beta inhibitor, and a MEK inhibitor promoted their differentiation into amniotic ectoderm. Using a co-culture technique to form cellular aggregates, he was able to confirm the ability of the generated amniotic ectoderm cells to initiate gastrulation. Also, single cell RNA sequencing analysis suggested the differentiation pathway for amniotic ectoderm, where surface ectoderm genes are expressed before the activation of the amniotic ectoderm gene programme.
Comparison of the gene expression data against public data from primate embryos at comparable developmental stages, performed with the expertise of Dr Irina Mohorianu, Head of Bioinformatics at the CSCI, validated the cell types generated in the culture system, demonstrating their similarity to the corresponding embryonic tissues. Moreover, the developmental pathway through surface ectoderm state to amniotic ectoderm state was also observed in primate embryos.
Vitally, Dr Nakanoh identified that the distinction between the two cell types depends on cell density; where cells become amniotic ectoderm in sparse conditions. In contrast, high density culturing led to cells that expressed markers for surface ectoderm but not amniotic ectoderm. These cells were also able to differentiate into a downstream epidermal cell type, keratinocytes, which represent a key cell type in the skin. The researchers tested variations in the culture media but cell density was the only factor that influenced the cell fate choice between amniotic ectoderm and surface ectoderm.
The research team also found that the new culture system produces cells like extraembryonic mesoderm, which is not yet well characterised in human embryos.
Dr Shota Nakanoh, a postdoctoral researcher in Affiliate PI Dr Theresa Rayon’s lab, said: “Amnion protects the embryo and provides key developmental cues while surface ectoderm contributes to a substantial part of adult body. Both cell types are of high clinical interest and are important elements for successful in vitro models for human embryos. Although there are protocols to differentiate cells to either amniotic or surface ectoderms, researchers were not able to make a clear distinction between these cell types. Our findings about cellular density as a key regulator fills this knowledge gap and thus facilitate us to be more certain about generating the cell types of interest.”
Applying this cell crowding effect back to the developing human embryo, the researchers propose how these similar tissues are differentiated in the pre-gastrulation embryo, where amniotic ectoderm arises as a loose sheet with relatively small numbers of cells, while surface ectoderm is formed as a continuous dense sheet of cells.
Enabling better stem cell embryo models
A method to correctly specify amnion formation is important to be able to recapitulate human embryo development as closely as possible using stem cell-based embryo models. These models are increasingly being used to explore human development beyond the technical and regulatory limits that govern human embryo research.
Dr Teresa Rayon, tenure-track group leader in Babraham Institute’s Epigenetics research programme, said: “We have only recently begun to explore the generation of amnion during human development. These findings advance our understanding on how to generate extra-embryonic cells in the lab in vitro, and sheds light on the mechanisms that drive the formation of cell types at the stages that correspond to the ‘black box’ of human development. Given the growing interest in using stem cell embryo models as proxies of human embryos, this work provides more knowledge for the generation of successful integrated models.”
Prof. Vallier, formerly of CSCI and now Professor of Stem Cells in Regenerative Therapies at the Berlin Institute of Health at Charité (BIH), said: “Our culture system also generates extra-embryonic mesoderm, another tissue not studied well in human embryos. It will provide better understanding of human development and could improve our knowledge about diseases affecting first step of foetal life. This work also opens the door for new studies regarding the role of cellular density in cell fate decision.”
