Cambridge stem cell scientists have developed a new zebrafish model to study the origins of Fanconi anemia, a paediatric disease that causes fatal bone marrow failure and predisposes children to cancer.
Using this model, the researchers showed the symptoms of this disease to be linked to genetic changes that disrupt blood stem cells in developing embryos. This new understanding, alongside the development of a novel research model, lays the foundations for further advancements in Fanconi anemia research and new drug discovery opportunities.
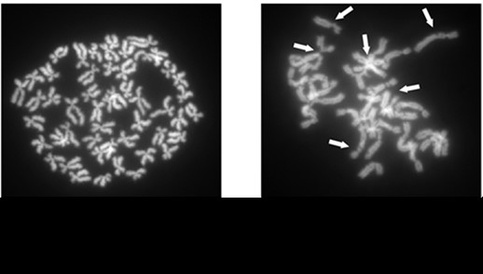
The research team, led by Dr Ana Cvejic from the Wellcome Trust – MRC Cambridge Stem Cell Institute, used genetically altered zebrafish to investigate RAD51, a gene recently discovered to be involved with Fancomi anemia and DNA repair. Their studies showed that fish without the RAD51 gene were hypersensitive to DNA damage, one of the key features of Fanconi anemia.
These genetically altered fish also showed symptoms similar to those seen in humans, such as decreased size and increased cancer incidence, as well as key haematological features of the disease. Through observation of the developing fish eggs, the scientists learned that these disease symptoms, normally diagnosed in childhood, are actually linked to changes in blood stem cells during embryonic development.
This new research appears in the journal Proceedings of the National Academy of Sciences, and has the potential to revolutionise research into Fanconi anemia, an inherited disease typically diagnosed in children aged 2 – 15 years old.
“Using fish from the Sanger Institute Zebrafish Mutation Project, we have developed an important new model to advance research into Fanconi anemia” explains Dr Cvejic. “We have investigated the gene RAD51, which was known to be involved in the development of this disease, however the specifics of the biology were unclear.
Our results show that changes in RAD51 drive dysfunction of blood stem cells during embryonic development which leads on to the typical bone marrow failure seen in children with Fanconi anemia. We are confident that further research using our model will increase understanding of Fanconi anemia, and will have significant impacts on the development of new therapeutics for people living with this disease.”
Dr Cvejic and her team are part of the Department of Haematology at the University of Cambridge, and are based at the Wellcome Trust Sanger Institute. The group will build on this new research and work towards their goal of fully understanding the biology of blood stem cells, paving the way for their full medical potential to be reached.
The continued support of the MRC, Wellcome and Cancer Research UK, as well as the European Hematology Association and the Isaac Newton Trust is gratefully acknowledged by Ana and her team.
Hear more from Ana Cvejic about her research in our #cscipodcast
Publication details:
Botthof J, Bielczyk-Maczynska E, Ferreira L, Cvejic A. Loss of the homologous recombination gene rad51 leads to Fanconi anemia-like symptoms in zebrafish. Proceedings of the National Academy of Sciences doi:10.1073/pnas.1620631114