Professor Ana Cvejic
Haematopoietic stem cells
Email: ana.cvejic@bric.ku.dk and as889@cam.ac.uk
Biography
Ana Cvejic is a Professor at the University of Copenhagen and a Group leader at the Biotech Research and Innovation Centre (BRIC), Denmark. In 2008 Ana received her PhD in Biochemistry at the University of Bristol. She then moved to the University of Cambridge/Wellcome Trust Sanger Institute to start a Postdoctoral Fellowship, with Professor Willem Ouwehand. In 2012 Ana was awarded the CRUK Career Development Fellowship to start her independent group at the University of Cambridge. In 2015 Ana was awarded ERC Starting Grant and in 2016 EMBO Young Investigator award. In 2022 Ana was a recipient of the ERC Consolidator Grant and she moved her group to the University of Copenhagen. With the principal expertise and research interest in the molecular regulation of blood stem cell fate choices Ana’s research sits at the intersection of molecular biology, genetics and systems biology and it closely couples experimental approaches and “big” biological data analysis.
Funding
European Research Council (ERC), MRC, Open Targets, Novo Nordisk Foundation
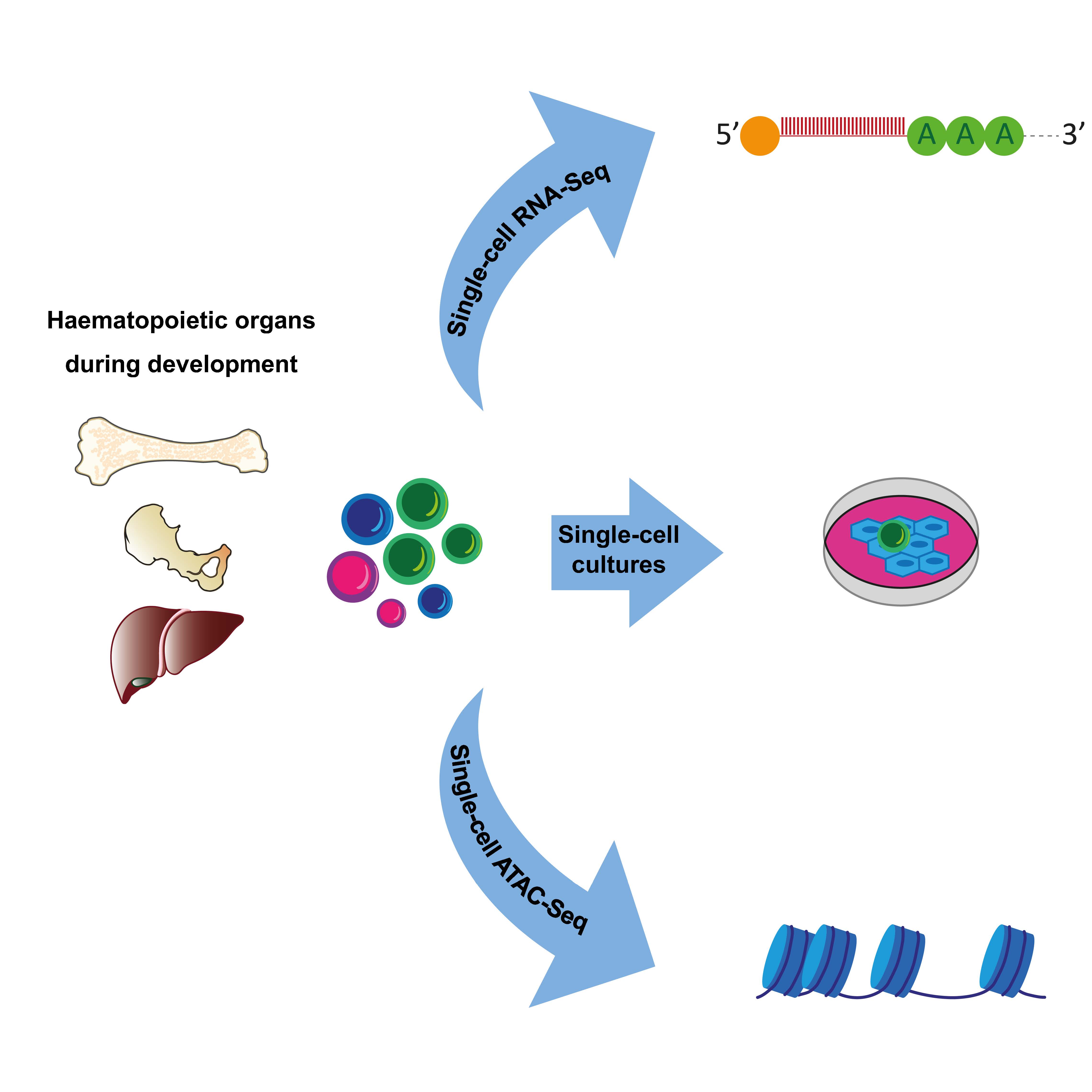
The Cvejic Group generated a detailed transcriptional and chromatin accessibility map of foetal liver and bone marrow haematopoietic stem cells (HSCs). Within HSCs, they revealed extensive epigenetic but not transcriptional priming. They also identified transcriptional and functional differences between HSC from liver and bone marrow.
Research
Blood stem cells need to both perpetuate (self-renew) themselves and differentiate into all mature blood cells to maintain blood formation throughout life. Clarifying how haemopoietic stem and progenitor cells (HSPCs) differentiate into diverse cell types is important for understanding how this process is subverted in the generation of blood pathologies.
The aim of the Cvejic group is to decipher how differentiation pathways of HSPCs are influenced by different microenvironments. To achieve that they use state-of-the-art single-cell RNA-seq data generation combined with computational analysis to establish principles of blood lineage differentiation. In particularly they are focusing on the dissection of the heterogeneity of cellular states in the blood system. The Group's research involves the use of both model organism (zebrafish, Danio rerio) and human samples. Currently, they are working with human foetal haematopoietic cells to reveal the dynamics and cellular programmes active during human blood development as well as lung cancer patient samples to investigating the influence of tumour microenvironment in the context of pathological differentiation of myeloid progenitors.
The results from their studies will advance our understanding of how normal fate decisions are instigated and provide clues for the design of novel therapies for blood pathologies.
Plain English
A single cell type, the haematopoietic stem cell (HSC), is responsible for generating all blood cells throughout the lifetime of an organism. The HSC is a rare cell that resides primarily in the bone marrow of adult mammals. It has the ability to either self-renew, and generate more stem cells or differentiate and generate over 10 different blood cell types. These different blood cells provide functions such as protection against infections, oxygen transport and maintaining haemostasis. Thus, over time each HSC makes essential fate decisions by integrating a wide array of signals from the microenvironment and completing complex changes in the regulation of gene expression. Clarifying how HSCs differentiate into diverse cell types is important for understanding how they attain their various functions and offers the potential for therapeutic manipulation.
Key Publications
- Spencer Chapman M, Ranzoni AM, Myers B, Williams N, Coorens THH, Mitchell E, Butler T, Dawson KJ, Hooks Y, Moore L, Nangalia J, Robinson PS, Yoshida K, Hook E, Campbell PJ, Cvejic A (2021). Lineage tracing of human development through somatic mutations. Nature. doi: https://doi.org/10.1038/s41586-021-03548-6
- Ranzoni AM, Tangherloni A, Berest I, Riva SG, Myers B, Strzelecka PM, Xu J, Panada E, Mohorianu I, Zaugg JB, Cvejic A (2020). Integrative Single-Cell RNA-Seq and ATAC-Seq Analysis of Human Developmental Hematopoiesis. Cell Stem Cell. 2020 Dec 15:S1934-5909(20)30553-1.
- Canu G, Athanasiadis E, Grandy RA, Garcia-Bernardo J, Strzelecka PM, Vallier L, Ortmann D, Cvejic A (2020). Analysis of endothelial-to-haematopoietic transition at the single cell level identifies cell cycle regulation as a driver of differentiation. Genome Biol. 2020 Jul 1;21(1):157. PMCID: PMC7329542
- Hernández PP, Strzelecka PM, Athanasiadis EI, Hall D, Robalo AF, Collins CM, Boudinot P, Levraud JP, Cvejic A (2018). Single-cell transcriptional analysis reveals ILC-like cells in zebrafish. Sci Immunol, 2018 Nov 16;3(29). pii: eaau5265. PMCID: PMC6258902
- Athanasiadis EI, Botthof JG, Andres H, Ferreira L, Lio P, Cvejic A. Single-cell RNA-sequencing uncovers transcriptional states and fate decisions in haematopoiesis. Nature Communications. 2017 Dec 11;8(1):2045. PMCID: PMC572549